Our research projects are highly interdisciplinary, combining chemistry, biology, physics, and material science. Our ultimate goal is the development of new strategies for controlled self-assembly of functional Nucleic Acid nanoparticles with implications in areas as diverse as nanoelectronics, bio-sensing, and nanomedicine. The list of projects below provide students with extensive training in Nucleic Acid 3D design and hands-on experience to master various biochemical and physicochemical techniques:
Nanobiotechnology
In this project, we implement three basic RNA nanotechnology approaches to construct functional and robust RNA nanoparticles. (i) Computational approach includes in silico (computer-aided) design of RNA nanoobjects (2D and 3D geometries) using naturally occurring RNA building blocks. (ii) Experimental approach includes RNA synthesis, self-assembly, and structural characterization of the designed RNA nanoparticles. (iii) In the functionalization approach, depending on the intended application, these nanoparticles can be decorated with a variety of biologically active moieties, such as cell-specific binding aptamers, silencing RNA, microRNA, small-molecule drugs, functional polypeptides, fluorescent probes, and more.
Bio-computing using Fluorogenic “Light-Up” RNA aptamers
The driving core of the fully operational computer system lies at the basis of Boolean logic gates and, therefore, to create a bio-computer one has to first generate biomolecular device possessing logic gate functons. Fluorogenic RNA and DNA aptamers are attractive candidates to fulfill this task due to their various light emitting properties and structural diversity.
In our lab, we implement secondary structure prediction algorithms to design and construct RNA and RNA/DNA complexes that are able to perform basic AND, OR, NOR, NAND logistic operations. Our goal is to achieve a nucleic acid based combinatorial logic system with unbiased emission output from a set of short oligonucleotide inputs.
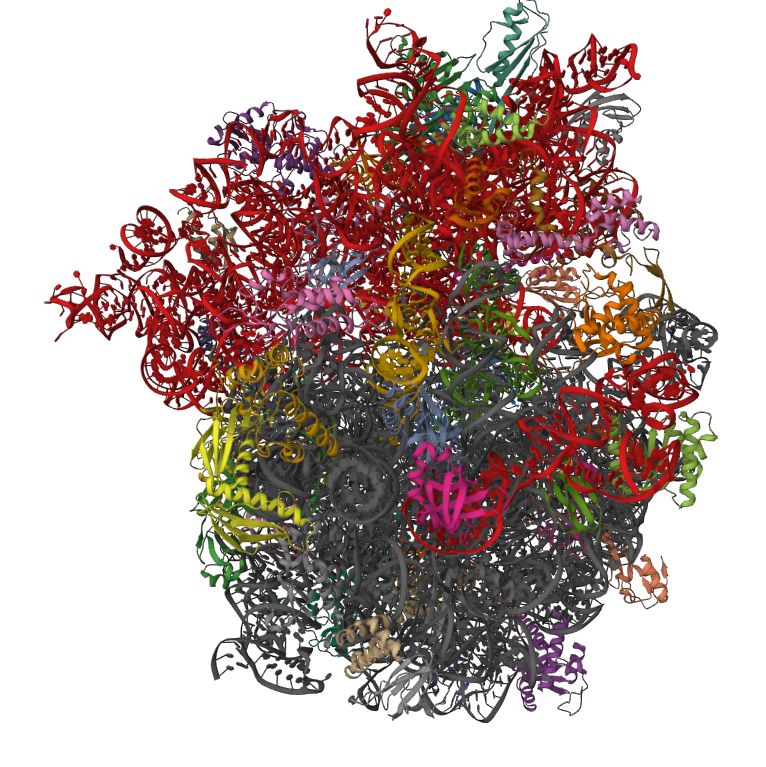
Thermodynamics of RNA non-canonical interactions
Structures of large RNA molecules, such as the ribosomal RNAs (rRNA), have revealed that no more than ~70% of all basepairs are cis Watson-Crick (cWW) AU, GC, or GU pairs. The rest are non-WC basepairs, and together with base-stacking and base-backbone interactions, they form single-stranded regions of hairpin loops, internal loops, and multi-helix junctions, many of which are recurrent.
Multiple nucleotides are isosteric, indicating that basepairs can potentially substitute for each other while preserving the 3D structure of the overall motif. However, only limited information is available about the relative effects of isosteric vs. non-isosteric base substitutions on the thermodynamic stabilities of well-structured RNA 3D motifs. This project addresses fundamental questions regarding the thermodynamic and structural stability of native RNA molecules.
We want to thank our sponsors for funding our research projects: